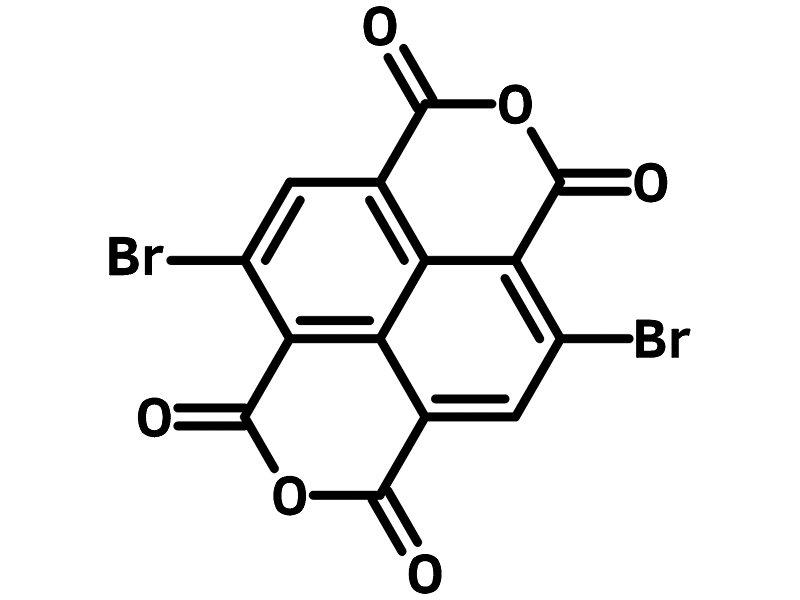
2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic Dianhydride
CAS Number 83204-68-6
Chemistry Building Blocks, Dibromo Monomers, Heterocyclic Building Blocks, Materials, Monomers2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic Dianhydride (NTCDA-2Br)
A building block for the synthesis of n-type semiconducting polymers and dyes for polymer solar cells, OFETs and bioimaging applications
Specifications | MSDS | Literature and Reviews
2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic dianhydride (NTCDA-2Br, CAS number 83204-68-6) is a symmetric naphthalene derivative with adjacent carboxylic acids forming anhydride on both ends of the structure. It is a precursor to naphthalene diimides (NDIs) which are used as active layer materials in organic solar cells, field-effect transistors.
2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic dianhydride possesses aromatic planar conjugated structure and is favourable for good charge transport. Materials composed of naphthalene-1,4,5,8-tetracarboxylic dianhydride (NTDCA) or naphthalene-diimides (NDIs) have an excellent combination of redox, electrical, optical and thermal properties with high moisture and oxygen resistance.
2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic dianhydride on its own could be used as cathode interlayer materials for efficient electron extraction.
Naphthalenecarboxylic dianhydride building block
for the synthesis of OLED and organic photovoltaic materials
Worldwide shipping
Quick and reliable shipping
Capped with bromides
for facile coupling reactions
High purity
>98% Purity
General Information
| CAS Number | 83204-68-6 |
| Chemical Formula | C14H2Br2O6 |
| Full Name | 2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic Dianhydride |
| Molecular Weight | 425.97 g/mol |
| Synonyms |
4,9-Dibromoisochromeno[6,5,4-def]isochromene1,3,6,8-tetraone, NTCDA-2Br |
| Classification / Family |
Naphthalenes, Monomers, Electron transport layer materials |
Chemical Structure
Product Details
| Purity | >98% (GC) |
| Melting Point | 350 °C |
| Appearance | Pale yellow to yellow powder |
MSDS Documentation
 2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic dianhydride MSDS Sheet
2,6-Dibromonaphthalene-1,4,5,8-tetracarboxylic dianhydride MSDS Sheet
Literature and Reviews
-
A Balanced Face-On to Edge-On Texture Ratio in Naphthalene Diimide-Based Polymers with Hybrid Siloxane Chains Directs Highly Efficient Electron Transport, Y. Kim et al., Macromolecules 48 (15), 5179–5187 (2015); DOI: 10.1021/acs.macromol.5b01012.
-
The Effect of Alkyl Spacers on the Mixed Ionic-Electronic Conduction Properties of N-Type Polymers, I. Maria et al., Adv. Funct. Mater., 31, 2008718 (2021); DOI: 10.1002/adfm.202008718.
-
The synthesis of novel core-substituted naphthalene diimides via Suzuki cross-coupling and their properties, S. Bhosale et al., New J. Chem., 33, 2409-2413 (2009); DOI: 10.1039/B9NJ00476A.